Chemistry of Single Use Materials
How does the chemical composition and structure of a substance determine its properties with
regard to its use, capacity for reuse, and capacity for recycling?
Chemistry plays an all important role in how a substance is created, used, recycled, and disposed of. The type of chemical bond that holds a single use material together directly determines how it is created and destroyed. A crushed aluminum can when recycled will be brought to it’s melting point and shaped back into another aluminum can because the aluminum atoms all share a sea of electrons and exist in no specific pattern. Aluminum is a breeze to recycle because of it’s pure metallic properties. LDPE and PVC are two plastics similar in structure and physical properties but different in chemical properties and therefore cannot be shredded and recycled together. Heating and molding that mixture will break chemical bonds and could render the whole batch of plastic useless.
How do the choices we make as consumers (purchasing, use, reuse, recycling, and discarding of materials) impact our local community and environment, and the global community and environment?
Single use materials can end up a few places when their lifespan is up. The first one is the landfill, where metals get buried and rust, paper decomposes into dirt, and plastics decompose slowly into one of two things. If not exposed to oxygen, the carbon bonds will break and reform surrounded by hydrogen. This makes methane, which is a flammable greenhouse gas with even more heat trapping capability than typical CO2. Otherwise, if left exposed to atmosphere, the carbon will bond with oxygen releasing CO2 and H2. In a landfill, it’s almost guaranteed that plastics and anything carbon based and decomposable will become methane because by the time it starts to degrade, it has already become buried by either dirt or more trash. This is why it’s best not to throw away plastics, and even more so anything that says it’s biodegradable. Biodegradable coffee cups, food waste, and other easily decomposable waste materials are best to be composted, where they can oxidize properly and generate a carbon and nitrogen rich fertilizer. When plastics are properly recycled, they get shredded, melted down, and dyed for reused by a manufacturer that accepts raw recycled material. This is possibly the best thing that can happen to them, however due to the recent economic climate, it’s become less sustainable and more energy intensive to re-use polymer plastics in this way.
What else did you learn through this project?
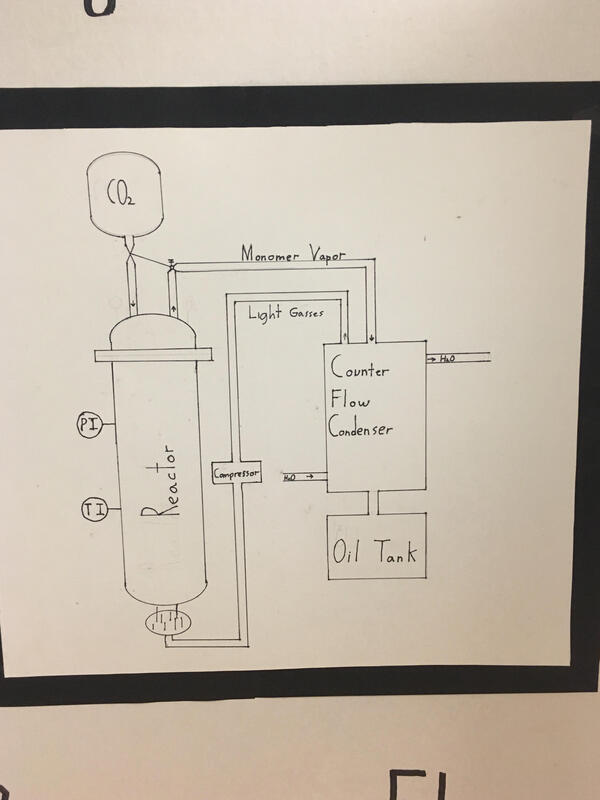
I enjoyed researching and learning about the process of pyrolysis. I had never thought it possible to reverse polymerization, as those are extremely tough chains of repeating molecules. When plastic melts the chains simply become loose, and when it gets hot enough it begins to catch fire, which is undesirable when it comes to reusing material. Pyrolysis bring polymers to a temperature high enough to break random bonds along long polymer chains. Normally this would result in oxygen bonding with both the carbon and hydrogen creating CO2 and H2O (Fire). Pyrolysis reactors take advantage of the reaction requiring oxygen by depriving the reaction chamber of such a gas. This allows the short, light, hydrocarbon chains to escape as a gas and re-condense as a liquid at a colder temperature. The product is a mix of oils that can be refined and separated into things like diesel and other natural gasses. The chains that are short enough to remain as a gas even after being cooled to room temperature are light gasses that can be used to fuel the reaction again, as they are not worth their weight and more useful as fuel on the spot. The process for me shines light on some of the simplicity to the chemical and industrial world.
regard to its use, capacity for reuse, and capacity for recycling?
Chemistry plays an all important role in how a substance is created, used, recycled, and disposed of. The type of chemical bond that holds a single use material together directly determines how it is created and destroyed. A crushed aluminum can when recycled will be brought to it’s melting point and shaped back into another aluminum can because the aluminum atoms all share a sea of electrons and exist in no specific pattern. Aluminum is a breeze to recycle because of it’s pure metallic properties. LDPE and PVC are two plastics similar in structure and physical properties but different in chemical properties and therefore cannot be shredded and recycled together. Heating and molding that mixture will break chemical bonds and could render the whole batch of plastic useless.
How do the choices we make as consumers (purchasing, use, reuse, recycling, and discarding of materials) impact our local community and environment, and the global community and environment?
Single use materials can end up a few places when their lifespan is up. The first one is the landfill, where metals get buried and rust, paper decomposes into dirt, and plastics decompose slowly into one of two things. If not exposed to oxygen, the carbon bonds will break and reform surrounded by hydrogen. This makes methane, which is a flammable greenhouse gas with even more heat trapping capability than typical CO2. Otherwise, if left exposed to atmosphere, the carbon will bond with oxygen releasing CO2 and H2. In a landfill, it’s almost guaranteed that plastics and anything carbon based and decomposable will become methane because by the time it starts to degrade, it has already become buried by either dirt or more trash. This is why it’s best not to throw away plastics, and even more so anything that says it’s biodegradable. Biodegradable coffee cups, food waste, and other easily decomposable waste materials are best to be composted, where they can oxidize properly and generate a carbon and nitrogen rich fertilizer. When plastics are properly recycled, they get shredded, melted down, and dyed for reused by a manufacturer that accepts raw recycled material. This is possibly the best thing that can happen to them, however due to the recent economic climate, it’s become less sustainable and more energy intensive to re-use polymer plastics in this way.
What else did you learn through this project?
I enjoyed researching and learning about the process of pyrolysis. I had never thought it possible to reverse polymerization, as those are extremely tough chains of repeating molecules. When plastic melts the chains simply become loose, and when it gets hot enough it begins to catch fire, which is undesirable when it comes to reusing material. Pyrolysis bring polymers to a temperature high enough to break random bonds along long polymer chains. Normally this would result in oxygen bonding with both the carbon and hydrogen creating CO2 and H2O (Fire). Pyrolysis reactors take advantage of the reaction requiring oxygen by depriving the reaction chamber of such a gas. This allows the short, light, hydrocarbon chains to escape as a gas and re-condense as a liquid at a colder temperature. The product is a mix of oils that can be refined and separated into things like diesel and other natural gasses. The chains that are short enough to remain as a gas even after being cooled to room temperature are light gasses that can be used to fuel the reaction again, as they are not worth their weight and more useful as fuel on the spot. The process for me shines light on some of the simplicity to the chemical and industrial world.